Histidine: Difference between revisions
No edit summary |
No edit summary |
||
| Line 2: | Line 2: | ||
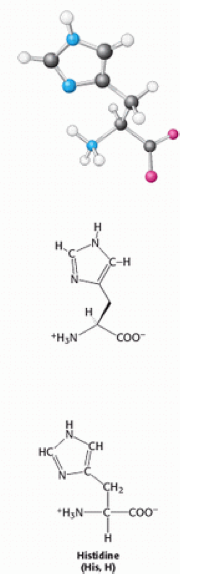
Histidine is an [[Aromatic_ring|aromatic]] amino acid, which acts as an [[acid|acid]] when [[Proton|protonated]]. When unprotonated it has a positively charged [[imidazole|imidazole]] functional group and acts as a general base. It plays an important role in stabilising the folding structures of proteins and active sites of [[enzymes|enzymes]] <ref>http://www.biology.arizona.edu/biochemistry/problem_sets/aa/histidine.html</ref> . This ability is due to its binding properties with metals and the basic properties of the nitrogen [[atoms|atoms]] in the ring. It is only found as an L- isomer and it plays an important role in growth and repair of tissues in the body. | Histidine is an [[Aromatic_ring|aromatic]] amino acid, which acts as an [[acid|acid]] when [[Proton|protonated]]. When unprotonated it has a positively charged [[imidazole|imidazole]] functional group and acts as a general base. It plays an important role in stabilising the folding structures of proteins and active sites of [[enzymes|enzymes]] <ref>http://www.biology.arizona.edu/biochemistry/problem_sets/aa/histidine.html</ref> . This ability is due to its binding properties with metals and the basic properties of the nitrogen [[atoms|atoms]] in the ring. It is only found as an L- isomer and it plays an important role in growth and repair of tissues in the body. | ||
[[Image:Histidine.png]] | |||
=== References === | === References === | ||
< | Berg, J. M., Tymoczko, J. L., & Stryer, L. (2002). Biochemistry (5th ed.). New York: W.H. Freeman.<br> | ||
Revision as of 14:41, 29 November 2013
Histidine (commonly abbreviated to His or H) [1] is one of the twenty naturally-occurring amino acids. Of these amino acids it is one of ten polar and one of three positively-charged amino acids [2]. The codons of His are CAU or CAC.
Histidine is an aromatic amino acid, which acts as an acid when protonated. When unprotonated it has a positively charged imidazole functional group and acts as a general base. It plays an important role in stabilising the folding structures of proteins and active sites of enzymes [3] . This ability is due to its binding properties with metals and the basic properties of the nitrogen atoms in the ring. It is only found as an L- isomer and it plays an important role in growth and repair of tissues in the body.
References
Berg, J. M., Tymoczko, J. L., & Stryer, L. (2002). Biochemistry (5th ed.). New York: W.H. Freeman.
- ↑ ^ IUPAC-IUBMB Joint Commission on Biochemical Nomenclature. "Nomenclature and Symbolism for Amino Acids and Peptides". Recommendations on Organic and Biochemical Nomenclature, Symbols and Terminology etc. Retrieved 2007-05-17.
- ↑ http://www.detectingdesign.com/images/Abiogenesis/Amino%20Acid%20Chart.jpg
- ↑ http://www.biology.arizona.edu/biochemistry/problem_sets/aa/histidine.html