Optical isomers: Difference between revisions
Jump to navigation
Jump to search
Added a picture |
Good edit. Moved the image. |
||
| Line 1: | Line 1: | ||
[[Image: | [[Image:Optical isomerism.GIF|right|Optical isomers are non-superimposable]] | ||
Optical isomers rotate the plane of plane polarised light by 90° clockwise or anticlockwise. Optical isomers will have a central, chiral carbon atom. Chirality depends on the central C atom having four different groups attached to it as this makes the molecule non-superimposabe on its mirror image (asymmetrical)<ref>This is a reference to fckLRhttp://www.chemguide.co.uk/basicorg/isomerism/optical.html</ref>. Optical isomers (enantiomers) rotate the plane of plane polarised light in opposite directions. | Optical isomers rotate the plane of plane polarised light by 90° clockwise or anticlockwise. Optical isomers will have a central, chiral carbon atom. Chirality depends on the central C atom having four different groups attached to it as this makes the molecule non-superimposabe on its mirror image (asymmetrical)<ref>This is a reference to fckLRhttp://www.chemguide.co.uk/basicorg/isomerism/optical.html</ref>. Optical isomers (enantiomers) rotate the plane of plane polarised light in opposite directions. | ||
| Line 7: | Line 7: | ||
=== References === | === References === | ||
<references /> | <references /> | ||
<span style="border-top-left-radius: 2px; border-top-right-radius: 2px; border-bottom-right-radius: 2px; border-bottom-left-radius: 2px; text-indent: 20px; width: auto; padding: 0px 4px 0px 0px; text-align: center; font-style: normal; font-variant-caps: normal; font-weight: bold; font-size: 11px; line-height: 20px; font-family: 'Helvetica Neue', Helvetica, sans-serif; color: rgb(255, 255, 255); background-image: url(data:image/svg+xml;base64,PHN2ZyB4bWxucz0iaHR0cDovL3d3dy53My5vcmcvMjAwMC9zdmciIGhlaWdodD0iMzBweCIgd2lkdGg9IjMwcHgiIHZpZXdCb3g9Ii0xIC0xIDMxIDMxIj48Zz48cGF0aCBkPSJNMjkuNDQ5LDE0LjY2MiBDMjkuNDQ5LDIyLjcyMiAyMi44NjgsMjkuMjU2IDE0Ljc1LDI5LjI1NiBDNi42MzIsMjkuMjU2IDAuMDUxLDIyLjcyMiAwLjA1MSwxNC42NjIgQzAuMDUxLDYuNjAxIDYuNjMyLDAuMDY3IDE0Ljc1LDAuMDY3IEMyMi44NjgsMC4wNjcgMjkuNDQ5LDYuNjAxIDI5LjQ0OSwxNC42NjIiIGZpbGw9IiNmZmYiIHN0cm9rZT0iI2ZmZiIgc3Ryb2tlLXdpZHRoPSIxIj48L3BhdGg+PHBhdGggZD0iTTE0LjczMywxLjY4NiBDNy41MTYsMS42ODYgMS42NjUsNy40OTUgMS42NjUsMTQuNjYyIEMxLjY2NSwyMC4xNTkgNS4xMDksMjQuODU0IDkuOTcsMjYuNzQ0IEM5Ljg1NiwyNS43MTggOS43NTMsMjQuMTQzIDEwLjAxNiwyMy4wMjIgQzEwLjI1MywyMi4wMSAxMS41NDgsMTYuNTcyIDExLjU0OCwxNi41NzIgQzExLjU0OCwxNi41NzIgMTEuMTU3LDE1Ljc5NSAxMS4xNTcsMTQuNjQ2IEMxMS4xNTcsMTIuODQyIDEyLjIxMSwxMS40OTUgMTMuNTIyLDExLjQ5NSBDMTQuNjM3LDExLjQ5NSAxNS4xNzUsMTIuMzI2IDE1LjE3NSwxMy4zMjMgQzE1LjE3NSwxNC40MzYgMTQuNDYyLDE2LjEgMTQuMDkzLDE3LjY0MyBDMTMuNzg1LDE4LjkzNSAxNC43NDUsMTkuOTg4IDE2LjAyOCwxOS45ODggQzE4LjM1MSwxOS45ODggMjAuMTM2LDE3LjU1NiAyMC4xMzYsMTQuMDQ2IEMyMC4xMzYsMTAuOTM5IDE3Ljg4OCw4Ljc2NyAxNC42NzgsOC43NjcgQzEwLjk1OSw4Ljc2NyA4Ljc3NywxMS41MzYgOC43NzcsMTQuMzk4IEM4Ljc3NywxNS41MTMgOS4yMSwxNi43MDkgOS43NDksMTcuMzU5IEM5Ljg1NiwxNy40ODggOS44NzIsMTcuNiA5Ljg0LDE3LjczMSBDOS43NDEsMTguMTQxIDkuNTIsMTkuMDIzIDkuNDc3LDE5LjIwMyBDOS40MiwxOS40NCA5LjI4OCwxOS40OTEgOS4wNCwxOS4zNzYgQzcuNDA4LDE4LjYyMiA2LjM4NywxNi4yNTIgNi4zODcsMTQuMzQ5IEM2LjM4NywxMC4yNTYgOS4zODMsNi40OTcgMTUuMDIyLDYuNDk3IEMxOS41NTUsNi40OTcgMjMuMDc4LDkuNzA1IDIzLjA3OCwxMy45OTEgQzIzLjA3OCwxOC40NjMgMjAuMjM5LDIyLjA2MiAxNi4yOTcsMjIuMDYyIEMxNC45NzMsMjIuMDYyIDEzLjcyOCwyMS4zNzkgMTMuMzAyLDIwLjU3MiBDMTMuMzAyLDIwLjU3MiAxMi42NDcsMjMuMDUgMTIuNDg4LDIzLjY1NyBDMTIuMTkzLDI0Ljc4NCAxMS4zOTYsMjYuMTk2IDEwLjg2MywyNy4wNTggQzEyLjA4NiwyNy40MzQgMTMuMzg2LDI3LjYzNyAxNC43MzMsMjcuNjM3IEMyMS45NSwyNy42MzcgMjcuODAxLDIxLjgyOCAyNy44MDEsMTQuNjYyIEMyNy44MDEsNy40OTUgMjEuOTUsMS42ODYgMTQuNzMzLDEuNjg2IiBmaWxsPSIjYmQwODFjIj48L3BhdGg+PC9nPjwvc3ZnPg==); background-color: rgb(189, 8, 28); background-size: 14px 14px; position: absolute; opacity: 1; z-index: 8675309; display: none; cursor: pointer; border: none; -webkit-font-smoothing: antialiased; top: 15px; left: 15px; background-position: 3px 50%; background-repeat: no-repeat no-repeat;">Save</span><span style="border-top-left-radius: 2px; border-top-right-radius: 2px; border-bottom-right-radius: 2px; border-bottom-left-radius: 2px; text-indent: 20px; width: auto; padding: 0px 4px 0px 0px; text-align: center; font-style: normal; font-variant-caps: normal; font-weight: bold; font-size: 11px; line-height: 20px; font-family: 'Helvetica Neue', Helvetica, sans-serif; color: rgb(255, 255, 255); background-image: url(data:image/svg+xml;base64,PHN2ZyB4bWxucz0iaHR0cDovL3d3dy53My5vcmcvMjAwMC9zdmciIGhlaWdodD0iMzBweCIgd2lkdGg9IjMwcHgiIHZpZXdCb3g9Ii0xIC0xIDMxIDMxIj48Zz48cGF0aCBkPSJNMjkuNDQ5LDE0LjY2MiBDMjkuNDQ5LDIyLjcyMiAyMi44NjgsMjkuMjU2IDE0Ljc1LDI5LjI1NiBDNi42MzIsMjkuMjU2IDAuMDUxLDIyLjcyMiAwLjA1MSwxNC42NjIgQzAuMDUxLDYuNjAxIDYuNjMyLDAuMDY3IDE0Ljc1LDAuMDY3IEMyMi44NjgsMC4wNjcgMjkuNDQ5LDYuNjAxIDI5LjQ0OSwxNC42NjIiIGZpbGw9IiNmZmYiIHN0cm9rZT0iI2ZmZiIgc3Ryb2tlLXdpZHRoPSIxIj48L3BhdGg+PHBhdGggZD0iTTE0LjczMywxLjY4NiBDNy41MTYsMS42ODYgMS42NjUsNy40OTUgMS42NjUsMTQuNjYyIEMxLjY2NSwyMC4xNTkgNS4xMDksMjQuODU0IDkuOTcsMjYuNzQ0IEM5Ljg1NiwyNS43MTggOS43NTMsMjQuMTQzIDEwLjAxNiwyMy4wMjIgQzEwLjI1MywyMi4wMSAxMS41NDgsMTYuNTcyIDExLjU0OCwxNi41NzIgQzExLjU0OCwxNi41NzIgMTEuMTU3LDE1Ljc5NSAxMS4xNTcsMTQuNjQ2IEMxMS4xNTcsMTIuODQyIDEyLjIxMSwxMS40OTUgMTMuNTIyLDExLjQ5NSBDMTQuNjM3LDExLjQ5NSAxNS4xNzUsMTIuMzI2IDE1LjE3NSwxMy4zMjMgQzE1LjE3NSwxNC40MzYgMTQuNDYyLDE2LjEgMTQuMDkzLDE3LjY0MyBDMTMuNzg1LDE4LjkzNSAxNC43NDUsMTkuOTg4IDE2LjAyOCwxOS45ODggQzE4LjM1MSwxOS45ODggMjAuMTM2LDE3LjU1NiAyMC4xMzYsMTQuMDQ2IEMyMC4xMzYsMTAuOTM5IDE3Ljg4OCw4Ljc2NyAxNC42NzgsOC43NjcgQzEwLjk1OSw4Ljc2NyA4Ljc3NywxMS41MzYgOC43NzcsMTQuMzk4IEM4Ljc3NywxNS41MTMgOS4yMSwxNi43MDkgOS43NDksMTcuMzU5IEM5Ljg1NiwxNy40ODggOS44NzIsMTcuNiA5Ljg0LDE3LjczMSBDOS43NDEsMTguMTQxIDkuNTIsMTkuMDIzIDkuNDc3LDE5LjIwMyBDOS40MiwxOS40NCA5LjI4OCwxOS40OTEgOS4wNCwxOS4zNzYgQzcuNDA4LDE4LjYyMiA2LjM4NywxNi4yNTIgNi4zODcsMTQuMzQ5IEM2LjM4NywxMC4yNTYgOS4zODMsNi40OTcgMTUuMDIyLDYuNDk3IEMxOS41NTUsNi40OTcgMjMuMDc4LDkuNzA1IDIzLjA3OCwxMy45OTEgQzIzLjA3OCwxOC40NjMgMjAuMjM5LDIyLjA2MiAxNi4yOTcsMjIuMDYyIEMxNC45NzMsMjIuMDYyIDEzLjcyOCwyMS4zNzkgMTMuMzAyLDIwLjU3MiBDMTMuMzAyLDIwLjU3MiAxMi42NDcsMjMuMDUgMTIuNDg4LDIzLjY1NyBDMTIuMTkzLDI0Ljc4NCAxMS4zOTYsMjYuMTk2IDEwLjg2MywyNy4wNTggQzEyLjA4NiwyNy40MzQgMTMuMzg2LDI3LjYzNyAxNC43MzMsMjcuNjM3IEMyMS45NSwyNy42MzcgMjcuODAxLDIxLjgyOCAyNy44MDEsMTQuNjYyIEMyNy44MDEsNy40OTUgMjEuOTUsMS42ODYgMTQuNzMzLDEuNjg2IiBmaWxsPSIjYmQwODFjIj48L3BhdGg+PC9nPjwvc3ZnPg==); background-color: rgb(189, 8, 28); background-size: 14px 14px; position: absolute; opacity: 1; z-index: 8675309; display: none; cursor: pointer; border: none; -webkit-font-smoothing: antialiased; top: 15px; left: 15px; background-position: 3px 50%; background-repeat: no-repeat no-repeat;">Save</span> | |||
Revision as of 20:31, 4 December 2016
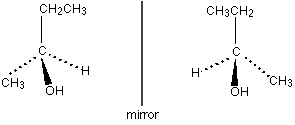
Optical isomers rotate the plane of plane polarised light by 90° clockwise or anticlockwise. Optical isomers will have a central, chiral carbon atom. Chirality depends on the central C atom having four different groups attached to it as this makes the molecule non-superimposabe on its mirror image (asymmetrical)[1]. Optical isomers (enantiomers) rotate the plane of plane polarised light in opposite directions.
Two enantiomers of an optical isomer will have very similar physical and chemical properties making it very hard to tell the difference between enantiomers.
References
- ↑ This is a reference to fckLRhttp://www.chemguide.co.uk/basicorg/isomerism/optical.html
SaveSave