Electron transfer chain
The electron transfer chain is a series of protein complexes; NADH-Q oxioreductase, Q-cytochrome C oxioreductase, cytochrome C oxidase and ATP synthase. In between these components are electrons carriers which will transfer the electrons from a complex to another. The protein complexes contain certain redox centres, and electrons move to carriers with increasing electron affinity along the chain: 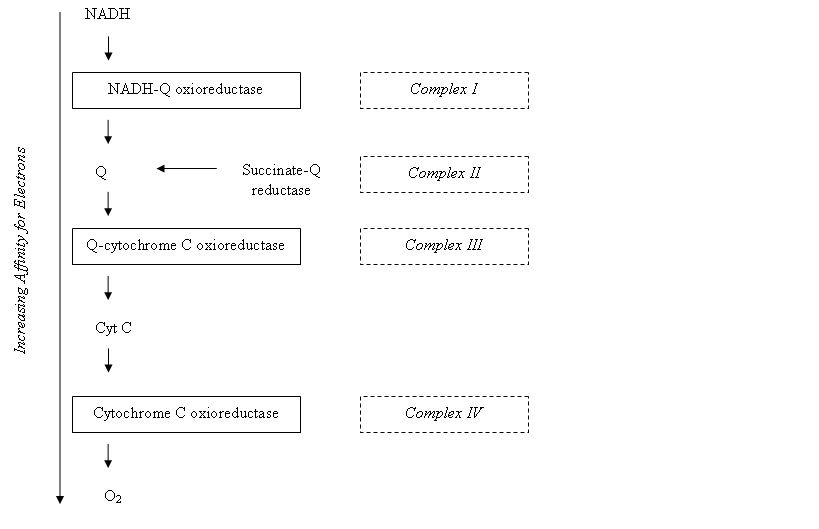
[Q is coenzyme Q, also known as ubiquinone, carries electrons from NADH and FADH2. It is hydrophobic and diffuses rapidly in the inner mitochondrial membrane.]
• Complex I – NADH-Q oxioreductase
o Transfer of two high-potential electrons from NADH to FMN
o Electrons from FMNH2 transferred to a series of Fe-S clusters
o Electrons from Fe-S clusters shuttled to Coenzyme Q
o Net Effect –
4H+ pumped out of matrix into intermembrane space
2 chemical H+ removed from matrix
• Complex II – Succinate-Q reductase complex
o Succinate dehydrogenase (in Cirtic Acid Cycle) part of succinate-Q reductase complex
o Electrons from FADH2 transferred to Fe-S clusters and then to Q
o Does not pump protons - therefore less ATP is formed by oxidation of FADH2 than NADH
• Complex III – Q-cytochrome C oxioreductase
o Cytochrome C is in all organisms with mitochondrial respiratory chains
o Small soluble protein containing C-type heme
o Carries one electron from Q-cytochrome C oxioreductase to cytochrome C oxidase
o Electrons transferred from QH2 to oxidised cytochrome C (CytCox)
o Mechanism that couples electron transfer is known as the Q Cycle
• Complex IV – Cytochrome C Oxidase
o 4 electrons transferred from cytochrome C to O2
o 4H+ from matrix allow the complete reduction of O2 to H2O
o 4 more H+ pumped across membrane
The electron motive force created by the flow of electrons down electron transport chain are used to pump H+ into the intermembrane space. This creates a concentration gradient between the inner membrane and the intermembrane space where the concentration of H in the intermembane space is higher than the concetration of H in the inner membrane space. As the inner membrane of the mitochondria is not permeable to H, this forces the H to go through the ATP synthase which will create a proton motive force to convert ADP + Pi into ATP.