CFTR
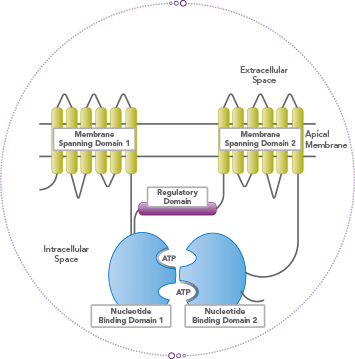
The Cystic Fibrosis Transmembrane Regulator protein (CFTR) regulates ion concentration in extracellular fluid due to its role as a plasma membrane Cl- channel on the lumenal (apical) membrane in epithelial cells [1]. It is a member of the ABC superfamily of proteins and is the only Cl- channel (passive transporter) in this family (the others are active transporters). It has got 12 transmembrane domains, with two nucleotide binding domains (NBD1 and NBD2) and a regulatory domain. The binding of ATP is essential for the function for the channel, NBD1 and NBD2 both bind ATP however only NBD2 can hydrolyse ATP. CFTR shares homology with P-glycoprotein, apart from the fact that CFTR contains a large regulatory domain (R domain).
The CFTR gene was cloned in 1989, and, when mutated, can cause cystic fibrosis. The mutation is recessive and therefore requires homozygous mutant alleles for the phenotype to manifest. So far, over 1600 mutations of the CFTR gene have been discovered, with the most common being the ∆F508(F598del) mutation, however some mutations have been identified as being unique to an individual. Some of the other more common mutations include; G542X G551D N1303K and W1282X.
In the ∆F508 mutation a phenylalanine residue at amino acid 508 has been deleted, leaving a sequence of only 1479 amino acids (formerly 1480). This mutation is a class II mutation where abnormal processing occurs in the endoplasmic reticulum. This abnormal processing means that the Nucleotide Binding Domain 1 (NBD1) cannot bind ATP correctly. Subsequent signalling from ATP binding to open/close the Cl- channel does not occur and the cell renders the channel non-functional [2]. The protein is then degraded via ubiquitin tagging [3]. The lack of Cl- channel means than Cl- cannot be transported out of the apical membrane into the lumen. Once Cl- accumulate in the cell, they remain both in the cell and on both sides of the basolateral membrane (the paracellular movement of Na+ is also decreased).
Cystic Fibrosis is a disease condition caused by several mutations in the gene encoding CFTR protein, which normally functions as a Cl- channel in the plasma membrane of epithelial cells. The CFTR channel plays a particularly important role, especially in the lungs, where it controls certain ion concentrations found in the extracellular fluid [4]. The disease causes decreased chloride concentration in the airway which leads to the thickening of the gel layer. As it thickens it becomes too heavy for the cilia to brush it along and so sticks to the cilia. Many organs become affected by Cystic Fibrosis, which disrupts their normal functioning. A major problem is encountered in the lungs, where there is clogging of the bronchial passage and infection. The repeated inflammation to combat infection damages lung tissue, so breathing is impeded and lungs are slowly destroyed. In the liver, disease affects digestion, as it plugs in the small bile ducts. In the pancreas, a precipitate forms in ducts to the bowel, meaning that digestive enzymes cannot be transported and digestion is insufficiently processed, leading to malnourishment. Other organs which are altered by the disease are the small intestine and the reproductive duct. In the small intestine thicky stool forms and clogs the ducts which sometimes even necessitates surgery, this is mostly seen in newborns. Females are occasionally affected by thick mucus stopping the passage of sperm to uterus thus making them infertile. Congenital absence bilateral absence of the vas deferens occurs in most of the males, when the vas deferens (testes) is absent or isn't properly formed, which results in infertility[5].
Currently, the main method of diagnosing cystic fibrosis is by analysing a sample of sweat from the patient. The eccrine cells are found all over the body and secrete a solution of sodium, chloride and water. Sodium chloride (NaCl) is reabsorbed from sweat through ENaC and CFTR channels in the epithelial cells of the eccrine duct as it travels towards the skin surface. As the duct is fairly impermeable to water, there is little osmosis; the solution that is eventually secreted from the lumen is hypotonic. In cystic fibrosis, CFTR is non-functional and so there is little chloride re-absorption at the apical membrane. The build up of the negative chloride charge in the duct impedes the diffusion of sodium. Sodium chloride is therefore trapped in the lumen and excreted in sweat, resulting in high ionic concentrations (>60mmol chloride concentration) in CF patients [6].
Treatment
There has not been any breakthrough yet. In vitro results have been sucessful however In vivo results shows very little cure rate. Phosphodiesterase type 5 inhibitors have been shown to restore the functioning of the ∆F508 CFTR gene in CF mice [7].
References
- ↑ Alberts, Bruce (2002), Molecular Biology Of The Cell, 4th edition, Garland Science
- ↑ Patrick AE and Thomas PJ (2012) Development of CFTR structure. Front. Pharmacol. 3:162. doi: 10.3389/fphar.2012.00162
- ↑ Alberts, Bruce (2002), Molecular Biology Of The Cell, 4th edition, Garland Science
- ↑ Alberts (2008) Molecular Biology of the Cell 5th ed. p.666. Garland Science
- ↑ Moskowitz SM, Chmiel JF, Sternen DL, et al. CFTR-Related Disorders. 2001 Mar 26 [Updated 2008 Feb 19]. In: Pagon RA, Adam MP, Ardinger HH, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2015. Available from: http://www.ncbi.nlm.nih.gov/books/NBK1250/
- ↑ [C. Schultz (2005) 'Defects of Secretion in Cystic Fibrosis', New York Springer]
- ↑ Noel S, Dhooghe B, Leal T. Front Pharmacol. 2012;3:167. Epub 2012 Sep 18.